How close are we to a COVID-19 vaccine?
Initial deliveries of the Oxford COVID-19 vaccine are expected within a matter of weeks, alongside a request to UK regulators for emergency use authorisation.

Oxford's ChAdOx1 nCoV-19 vaccine is widely believed to be the front running candidate for a vaccine against COVID-19. The vaccine was created by the University of Oxford's Jenner Institute and is being trialed with the help of National Health Service (NHS) Trusts in the UK. British-Swedish firm AstraZeneca has been granted exclusive rights for the vaccine (with commitments to ensure equitable global access and not make profit during the pandemic itself) - marketed as AZD1222.
Availability of the vaccine is dependant on two things; supply of the vaccine and approval on the basis of the upcoming Phase III trial results. With manufacture of the vaccine well underway and late-stage trials getting closer to delivering an answer, a COVID-19 vaccine may be closer than we think.
Delivery of the Oxford vaccine is set to begin in a matter of weeks with final "fill-and-finish" stages to start in September 2020 for delivery in October/November. As vaccine researchers learn about the efficacy of the vaccine, the UK Phase III trial has quietly been towards testing more on the elderly and use of booster doses. As final stage preliminary results are aggregated from trial sites around the world; we can expect to see an application for emergency use authorisation submitted to UK regulators submitted between late October and early November.
Whilst approval could be obtained even quicker by rapidly publishing efficacy data alone, given the need to ensure global equitable access and give increased priority to those who are at-risk, it is essential that both dosage and safety testing is thoroughly completed for at-risk groups for the vaccine to have maximum positive impact.
Supply of the Vaccine
Various governments around the world have made orders; these include orders of 100 million doses from the UK Government (30 million of which are to be delivered in the fall), followed by orders from the US Government, Europe’s Inclusive Vaccines Alliance (with a further order by the Swedish Government) and Australia. India's Serum Institute plans to make a billion doses in a year and will be marketed as "Covishield". Licensing and production deals have already been signed with companies in Russia and China.
There is a final step in the manufacturing process which is usually a bottleneck, the "fill-and-finish" stage. On the 3rd August 2020, the UK Government announced they had reached agreement with Wockhardt to use a facility in North Wales for the final "fill-and-finish" stage of a vaccine, which is expected to start in September 2020.
Equitable access has also been an important issue and the UK Government has already pledged equitable global access to coronavirus vaccines and treatments, including the Oxford vaccine. On June 4th, UK Prime Minister, Boris Johnson held an international fundraiser achieving $8.8 billion for the Gavi vaccine alliance (with the UK remaining the largest donor) to vaccinate 225 million children in low/middle income countries for various diseases, saving more than 700,000 lives. In March 2020, the British Government provided £210m funding to CEPI (the Coalition for Epidemic Preparedness Innovations), becoming their largest donor. In agreements with AstraZenecca, Gavi and CEPI's COVAX facility aims to supply 300 million doses with the Serum Institute of India suppling a billion doses for low/middle income countries.
Key to the partnership between Oxford researchers and the Jenner Institute was that AstraZenecca would manufacture such doses at-risk such that they can be deployed close to when/if the vaccine is proven to work, instead of needing to ramp up production then.
On the 7th June 2020, Bloomberg reported that AstraZenecca approached Gillead Sciences about considering a merger with the US firm. On the 23rd June, the UK Government changed merger rules by ministerial order to allow Government intervention "for the purpose of maintaining in the United Kingdom the capability to combat, or to mitigate the effects of, public health emergencies", giving the Government unilateral control of this matter whether under the standard, special or European intervention schemes.
Publication of Results
Recall when the NHS/Oxford RECOVERY trial found dexamethasone was an effective treatment for COVID-19 patients on ventilation or respirators, they initially made results available by a press release, then a pre-print paper which was ultimately published in a scientific journal (the full story on this is in my last blog post). In this instance, these results were essential to giving the doctors the knowledge to immediately begin saving lives around the world. By contrast, knowledge of vaccine studies offers no immediate benefit without regulatory approval and supply of the vaccine.
Oxford vaccine researchers have demonstrated their commitment to following rigorous scientific process instead of producing timely press releases. The Phase I/II results of the Oxford vaccine came out via a peer-reviewed scientific journal instead of via a pre-print or press release. It has also been reported that Sarah Gilbert, Professor of Oxford's Jenner Institute who is leading this vaccine effort, "appears to regard public attention as a distraction".
Progress
Vaccine development involves three phases of testing, Oxford's vaccine was the first to enter the Phase III stage - this is a large scale test which involves a placebo group and a vaccinated group which volunteers are randomised into. Efficacy of the vaccine is measured by comparing the two groups to see if there's a decrease in cases amongst the vaccinated group when compared to the placebo group. A Phase III trial also involves a large-scale safety study, including participants from more diverse backgrounds (such as the elderly) to see if the vaccine is suitable for them.
The first Phase III trial started in the UK on the 28th May 2020, is anticipated to involve a total of 12,330 participants and is still recruiting. Researchers initially hoped to gain results quickly, but as COVID-19 circulation reduced in the UK, it was clear it'd take longer than anticipated to get an efficacy signal.
On the 6th August 2020, vaccination of further participants was halted and 18-55 year old volunteers in London were told: "it is unlikely we will have an opportunity to vaccinate more participants of your age range on this particular trial". Trials continue with older patients, with even Andrew Lloyd Webber taking part.
If a study is closed prior to recruitment closing, this is a potential indication of the Haybittle–Peto boundary being met, where results look so promising that the trial can be completed early. If COVID-19 infection rates are similar amongst those in the vaccine study to those in the general population, we are roughly at the time where we should begin to see potential results coming from the UK Phase III study, which should allow research results to be unblinded for the researchers to identify efficacy.
On the 12th August 2020, the trial protocol was formally updated to version 4, with a greater focus on testing the number of doses that should be used, comparing a single dose against a Prime-Boost dose (where a participant receives one dose and then a booster dose later). This can be particularly useful in the elderly groups or those with weak immune responses (indeed, a trial in South Africa is testing the Prime-Boost regime of the Oxford vaccine in patients with and without HIV). The reasons for this are indicated in version 9 of the patient information sheet for 18-55 year olds (dated 20th July 2020):

As a backstop, since the 19th June 2020, a further Phase III study is already underway in Brazil which researchers expect to report preliminary data by November 2020. Another Phase I/II trial is underway in South Africa since the 23rd June 2020, which can help provide much needed efficacy data. A further study of 30,000 participants is planned in the US too with participants now being recruited. Most recently, the Serum Institute of India have also begun a Phase II/III trial of the same vaccine.
Safety Profile
Whilst it is true that some vaccine technologies like mRNA have not yet been used before, for example, in Moderna's mRNA-1273 vaccine - this is certainly not the case for the Oxford vaccine. The ChAdOx1 vector used in the Oxford vaccine has been tested in thousands of people, with published results dating back to 2014.
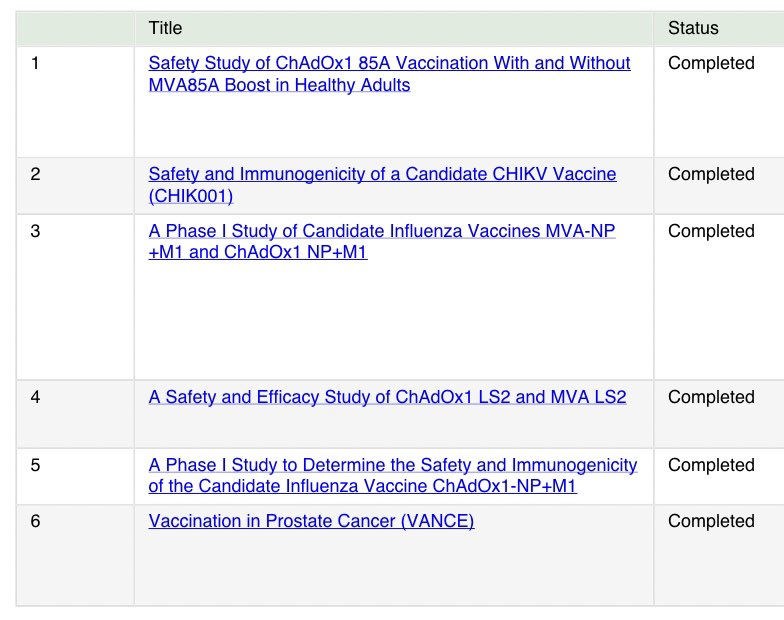
This is the key to how Oxford's Jenner Institute has been able to produce a vaccine so rapidly. The ChAdOx1 vector has been well-tested over a long period of time, used in many different ways for many different diseases. Professor Sarah Gilbert own 21 year-old triplets, are all taking part in the study of the vaccine their mother is leading the development of.
The Phase I/II results show no serious side effects of the Oxford vaccine despite despite a higher reactogenicity profile than a control meningitidis vaccine (indeed, the researchers identified a suspected unexpected serious adverse reaction with the meningitidis but there were no such issues with the COVID-19 vaccine).
The vaccine was well tolerated. Fatigue and headache were the most commonly reported systemic reactions, other reactions included muscle ache, feeling unwell and feeling feverish. Beside fatigue, these reactions were reduced in a group taking prophylactic paracetamol. The researchers found the use of prophylactic paracetamol increase tolerability and could reduce confusion with COVID-19 symptoms without needing to reduce dosage.
For the Oxford vaccine, neutralising antibody responses were found in 91%/100% of participants (depending on measurement approach) of those who took one dose but in 100% of participants following a booster dose.
By contrast; a similar vaccine developed by a Chinese firm, CanSino Biologics, was tested with both the same dose as the Oxford vaccine and a dose 2 times higher. Neutralising antibodies were only detected in 47% of participants with the same dose as the Oxford vaccine and in 59% in the higher dose. Due to safety concerns, the researchers only suggest proceeding with the lower dose despite a very limited immune response.
The key to why Oxford's ChAdOx1 vaccine seems far more effective is in the underlying vector used. CanSino Biologics uses a human adenovirus which humans can gain immunity to, indeed their study found 52% had high pre-existing immunity to the underlying vaccine vector before the trial even started.
By contrast, Oxford's vaccine uses the ChAdOx1 vector (Chimpanzee Adenovirus Oxford 1) is an animal adenovirus which humans don't have pre-existing immunity towards and there isn't a concern of developing immunity to the vector during the vaccination process either, allowing booster doses to take place and the vector to be used in future vaccines too.
Summary
So far, so good. The Oxford vaccine has been shown to be safe and produce a strong immune response, both with T-Cells and antibodies. Supply agreements around the world are in place and manufacturing is underway.
Based on combined trial data from multiple trial sites around the world; an application for emergency use authorisation to the UK MHRA (Medicines and Healthcare products Regulatory Agency) is expected to be submitted between late October and early November (8-12 weeks away), with deployment followed shortly after. To ensure the UK is able to maintain its commitment to equitable global access, priority will likely be given to frontline health and social care workers and those at increased risk.